SYBR Green based real-time PCR과 two-step RT-PCR을 신속히 수행할 수 있습니다
Products

QuantiFast SYBR Green PCR Kit (2000)
Cat. No. / ID: 204056
Features
- 60%까지 시간을 절약할 수 있는 보다 빠른 결과
- One protocol for all standard and fast cyclers
- Low copy에서도 specific detection이 가능
- 광범위한 template에서의 정확하게 검출이 가능함
- 최적화된 master mix와 protocol
Product Details
Performance
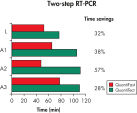
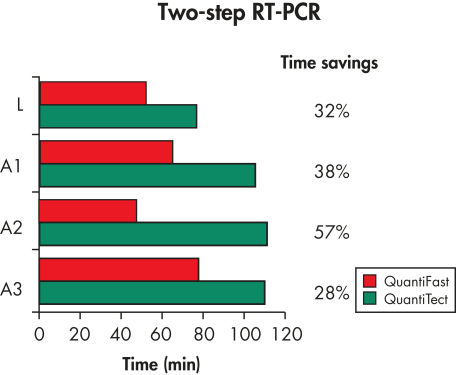
The QuantiFast SYBR Green PCR Kit delivers highly specific and sensitive results, outperforming other real-time PCR kits used in fast cycling mode (see figure " Sensitive two-step RT-PCR"). PCR run times are reduced by up to 60% (see figure " Significantly reduced PCR times"), allowing you to get results faster without compromising PCR performance (see figure " Faster results without compromising sensitivity"). You can also greatly increase your sample throughput or efficiently share a cycler with other users.
The QuantiFast SYBR Green PCR Kit enables accurate quantification of targets over several log dilutions of template. Even small differences in the amount of low-copy targets can be clearly distinguished (see figure " Resolution of small differences in copy number").
See figures
Principle
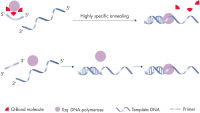
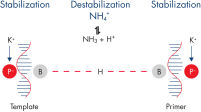
The QuantiFast SYBR Green PCR Kit delivers highly sensitive and specific results over a wide dynamic range on both standard and fast cyclers. The fluorescent dye SYBR Green I in the master mix enables the analysis of many different targets without having to synthesize target-specific labeled probes. A specially developed fast PCR buffer contains the novel PCR additive Q-Bond, which significantly reduces denaturation, annealing, and extension times (see figure " Fast primer annealing"). A balanced combination of K+ and NH4+ ions in the PCR buffer promotes specific primer annealing and enables high PCR specificity and sensitivity (see figure " Specific primer annealing"). In addition, HotStarTaq Plus DNA Polymerase requires only 5 minutes at 95°C for activation and provides a stringent hot start, preventing the formation of nonspecific products.
| Component | Features and benefits | Benefits |
|---|---|---|
| HotStarTaq Plus DNA Polymerase | 5 min activation at 95ºC | Set up of qPCR reactions at room temperature |
| QuantiFast SYBR Green PCR Buffer | Balanced combination of NH4+ and K+ ions | Specific primer annealing ensures reliable PCR results |
| Unique Q-Bond additive | Faster PCR run times, enabling faster results and more reactions per day | |
| SYBR Green I dye | Yields a strong fluorescent signal upon binding to double-stranded DNA | Highly sensitive amplification |
| ROX dye | Normalizes fluorescent signals on Applied Biosystems and, optionally, Agilent instruments | Precise quantification on cyclers that require ROX dye. Does not interfere with PCR on any real-time cycler |
See figures
Procedure
The QuantiFast SYBR Green PCR Kit is a ready-to-use master mix that eliminates the need for optimization of reaction and cycling conditions. Simply add primers and DNA template to the ready-to-use PCR master mix, and start the reaction. Follow the protocol in the handbook to get fast and reliable results on any real-time cycler.
For optimal results in real-time two-step RT-PCR, we recommend synthesizing cDNA using the QuantiTect Reverse Transcription Kit. The kit provides fast cDNA synthesis in just 20 minutes with integrated removal of genomic DNA contamination.
We also recommend QuantiTect Primer Assays for gene expression analysis using SYBR Green. QuantiTect Primer Assays are bioinformatically validated primer sets for any gene from human, mouse, rat, and many other species. Assays can be easily ordered online at the GeneGlobe Web portal.
Applications
The QuantiFast SYBR Green PCR Kit is for use in gene expression analysis of cDNA targets and quantitative gDNA analysis. QuantiFast SYBR Green PCR Kits are compatible with all available real-time cyclers, including instruments from Applied Biosystems, Bio-Rad, Cepheid, Eppendorf, Roche, and Agilent. For the Rotor-Gene Q and other Rotor-Gene cyclers, we recommend using the Rotor-Gene SYBR Green PCR Kit, which has been specially developed for fast cycling on these instruments.
Supporting data and figures
Significantly reduced PCR times.
Specifications
| Features | Specifications |
|---|---|
| Applications | SYBR Green-based, real-time PCR, two-step RT-PCR |
| Real-time or endpoint | Real-time |
| Sample/target type | cDNA, DNA |
| With or without ROX | With ROX |
| SYBR Green I or sequence-specific probes | SYBR Green I |
| Thermal cycler | Applied Biosystems, Bio-Rad, Cepheid, QIAGEN, Eppendorf, Roche, and Agilent |
| With/without hotstart | With hotstart |
| Single or multiplex | Single |
| Reaction type | Real-time and two-step RT-PCR |